Extracorporeal membrane oxygenation (ECMO) use in combat is usually reserved for a Critical Care Air Transport Teams (CCATT) like team. These ECMO teams are specially trained to perform this skill. As as military medic, you may find yourself assisting or eventually part of the care team for a patient on ECMO. Like many other medical advancements ECMO has a rocky history. The notion that the blood could be oxygenated through a membrane was discovered in the 1940s. In the 1970s there was a study on ECMO, however it was flawed due to variety of illnesses among the patients, the patients were often entered too late and already had irreversible damage, and vent support continued, which further caused damage. Eventually, ECMO was tried on children and the idea that it could correct lung damage was shown to be effective.
ECMO is used in military medicine for acute lung injury (ALI) and acute respiratory distress syndrome (ARDS). Both of these are the result of severe combat injuries, in fact, in critically wounded combat casualties, 26% had ALI and 33% had ARDS.
The Berlin definition of ARDS, is impaired oxygenation caused by acute, diffuse, inflammatory lung injury. However, to diagnose ARDS the following four criteria must be met:
- Acute disease onset
- Bilateral pulmonary infiltrates on chest X-ray
- Pulmonary capillary wedge pressure <18mmHg or absence of clinical evidence of left atrial hypertension
- Ratio between arterial oxygen partial pressure (PaO2) and the fraction of inspired oxygen (FiO2) <200
ALI is not as severe as ARDS and in order to diagnose it, the following criteria must be met: if they meet criteria 1-3 of ARDS, but exhibit a PaO /FiO ratio >200 and <300.
There are several factors that can contribute to ARDS or ALI, IEDs causing injuries to the lungs (primary blast injury, pulmonary contusion), high injury severity score, and blood transfusion. Here we will focus on ARDS.
ARDS results from inflammation-associated increases in pulmonary vascular permeability, edema and epithelial cell death. The histologic manifestation of these diseases is diffuse alveolar damage (DAD). It all starts with pneumocytes and the pulmonary endothelium. Just like the lethal triad, there is a cycle that results in significant damage and if untreated, will lead to death. There are four steps to the pathology of ARDS. From Robbins and Cotran Pathologic Basis of Disease:
- Endothelial activation: this is important early in the process. It activated by pneumocyte injury, which attracts the alveolar macrophages. In response, the macrophages secrete chemicals that act on the endothelium. Another means of activation is by circulating inflammatory chemicals. Some of these chemicals can cause injury to endothelial cells, while others activate the endothelial cells to express increased levels of adhesion molecules and procoagulant proteins.
- Adhesion and extravasation of neutrophils. Neutrophils stick to the endothelium and move into the interstitium and the alveoli. Once there, they degranulate and release inflammatory chemicals. Macrophage migration inhibitory factor is released and it helps to sustain the ongoing pro-inflammatory response. The result is increased recruitment and adhesion of leukocytes, causing more endothelial injury, and local thrombosis. This cycle of inflammation and endothelial damage lies at the heart of ALI/ARDS.
- Accumulation of intra-alveolar fluid and formation of hyaline membranes. The pulmonary capillaries become leaky secondary to the endothelial activation and injury, this results in fluid accumulation. Damage and death of the type II alveolar pneumocytes leads to surfactant issues, which further compromise alveolar gas exchange. Ultimately, the protein-rich fluid and debris from dead alveolar epithelial cells organize into hyaline membranes.
-
Resolution of injury is impeded due to the impaired ability of the remaining healthy cells to assist with edema resorption. Eventually, the inflammatory stimulus decreases, macrophages remove intra-alveolar debris and release specific chemicals. These chemicals stimulate fibroblast growth and collagen deposition, leading to fibrosis of alveoli and the walls. Pneumocytes are replaced by bronchiolar stem cells, which then proliferate. There is restoration through the proliferation of uninjured capillary endothelium.
ARDS will show by day six and should begin to resolve by day fourteen. A casualty may present with severe dyspnea and tachypnea, followed by cyanosis and hypoxemia (refractory to supplemental O2), and eventually respiratory failure. If x-ray is available, there will be diffuse bilateral infiltrates.
What is ECMO and how does it work? ECMO works by circumventing the heart and lungs, oxygenating the blood, and dispensing of the carbon dioxide and other waste that may be blown off by the respiratory system.
There are several types of ECMO, but most likely, you will see two in the military on combat casualties.
- Veno-arterial ECMO (vaECMO), a central vein is used to extract the blood which then goes to the ECMO machine, and a central artery is cannulated to return blood to the body. This type can be used for both cardiac and respiratory failure. The common site of access is the femoral vein and femoral artery.
- Veno-venous ECMO (vvECMO) is more common in the combat casualty. An advantage of vvECMO is, there is no arterial cannulation, therefore complications secondary to arterial embolic events are none. The venous cannulation is typically done percutaneous, whereas the arterial cannulation is performed surgically. When the ECMO is done through two sites, the preferred sites for cannulation are the femoral artery and the right internal jugular vein. When single site ECMO is done, the right internal jugular vein is preferred.
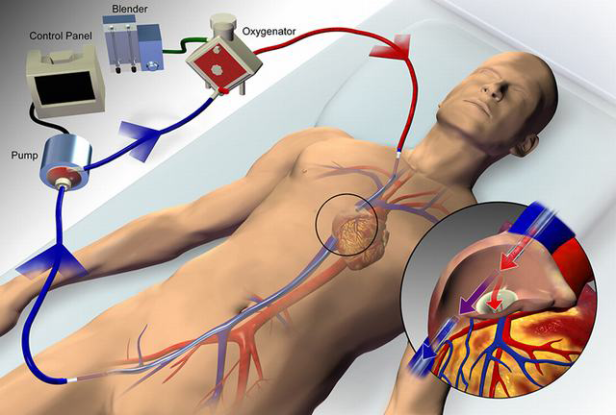
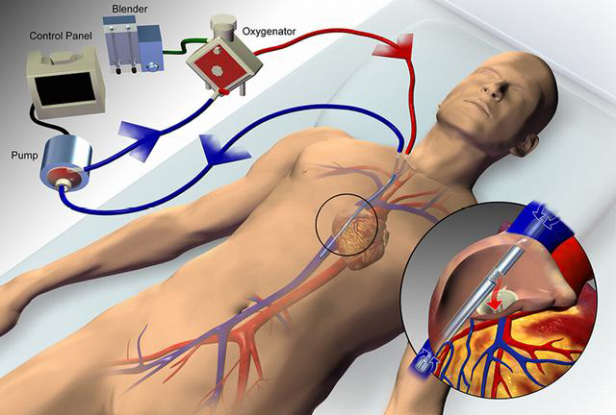
In vvECMO, a mixture of oxygen and air is connected to the membrane oxygenator, with the fraction of delivered oxygen set via a gas blender. This gas, referred to as sweep gas, passes along the side of a semipermeable membrane, while blood flows along the other side, the membrane allows for diffusion of O2 and CO2. The rate at which O2 moves into the blood include the amount of blood flow through the circuit relative to cardiac output, fraction of delivered oxygen through the circuit, the contribution of native lung gas exchange, and the characteristics of the membrane lung. Similar to how the aveoli/capillary blood flow determine CO2 removal, the rate of the sweep gas is the main determinant of CO2 removal. It can be titrated to PaCO2 or pH.
Casualties still need ventilatory management while on ECMO. The standard ventilator settings during ECMO is low-volume, low-pressure, the best settings for ARDS/ECMO is not really known. However, some experts say that extubation may be beneficial in ECMO. They will also need anticoagulation with heparin throughout their course on ECMO, but there is support for those with severe trauma to delay heparin for 24-72 hours.
The only multicenter randomized controlled study was the Conventional Ventilation or ECMO for Severe Adult Respiratory Failure (CESAR) trial. It consisted of 180 patients randomized into conventional management (n=90) or ECMO (n=90). The results demonstrated 63% (57/90) of the ECMO patients survived to 6 months compared with 47% (41/87) of the conventional management patients (p=0.03). Their conclusions stated that patients with a high Murray score (another ARDS scoring system) >3, they should be transferred to an ECMO-based management protocol to significantly improve survival without severe disability.
There is still a high mortality rate, varying sources report mortality from severe ARDS with ECMO is 30%-50%.

Is there room for ECMO in the prehospital/Role I setting? Is it possible that the technology will become portable enough to put in the PFC setting? Can the military medic learn the necessary skills to to ECMO? Right now, it’s not possible, however, many skills were once thought to difficult for the prehospital setting and the military medic. The technology is not ready and there is not enough data to support its use outside the hospital setting. Till the time when it may happen, be armed with ECMO knowledge, you never know when you may find yourself helping a severely wounded casualty who is on ECMO.

Hello, I am a paramedic with an acute interest in ventilation … is there any chance that the diffuse injuries seen in the casualties may be accidentally caused by the BVMR? if these casualties are mechanically ventilated and take baro trauma, could that explain some of the diffuse lung injury?
LikeLike