Note: types and dosages of medication used to treat altitude illness will be determined by your organization’s guidelines as authorized by your physician oversight. While this article does list medication doses, refer to your local treatment protocols for all care.
You and your team are conducting patrol operations on Mount Rainier (14,411 ft summit), a familiar route for your unit, as part of a training exercise. One of your teammates is new to the unit and it’s his first trip. As any young stud would do, he stayed up late drinking at the local bar the night before the climb and only got a few hours of sleep. The training event was supposed to be a steady climb to about 9,500 ft above sea level. However, with the surprise visit from the Sergeant Major and timelines getting tight, the First Sergeant says that you’re going to have to push that pace.
As time goes on and altitude increases your new teammate complains of a headache and dry mouth. You heard he was out drinking last night and scold him for being dehydrated and weak. The hard pace continues, and the steady ascent is not so steady. At about 8,800ft above sea level, he really looks like he’s dragging ass. He comes back to you with difficulty breathing, significant fatigue, and a non-productive cough! Your medic senses begin tingling and you realize something is just not right. What is causing this? Is he just the weak new guy? Is a bad hangover the culprit? Could this be caused by other factors?
Rapid ascent to a higher altitude can cause a number of physiologic stresses on the body ranging from mild headaches to life-threatening swelling in the lungs or brain. Prevention and early recognition are the two best treatments for any patient experiencing an altitude related illness.

How Does This Affect Me?
We’ve been operating in the mountains of Afghanistan for over 17 years. Our ability to move troops from the low altitude Piedmont of North Carolina and other similar CONUS installations to remote firebases at high altitude in a matter of days puts our Soldiers at a pretty decent risk of developing these kinds of illnesses. By knowing more about it, you can effectively treat or prevent this stuff from happening.
This article also gives you a jump start on Task 081-833-0035 “Initiate Treatment for High Altitude Illness” in MEDIC Table VI.
What is Altitude Illness?
First, let’s define what high-altitude is. The definition will vary depending on which book you read, but generally speaking*:
- High Altitude is 5000-11,500 feet (1500-3500 meters)
- Very High Altitude is 11,500-18,000 feet (3500-5500 meters)
- Extreme Altitude is anything above 18,000 feet (5500 meters)
* Conversions between Freedomheit and what the rest of the world uses have been fudged for the sake of clarity. Put your calculators away, nerds.
Altitude illness can be potentially lethal it isn’t recognized or if you lack the ability to treat it. As an example, the mountainous country of Nepal in South Asia where you will find such beasts as Mount Everest, records 2-3 deaths per week of tourists who failed to acclimatize.
Most of the problems people experience at high altitude are related to the lower pressure of the atmospheric. While the percentage of oxygen stays around 21%, the space between oxygen molecules is bigger. This is known as diminished partial pressure of inspired oxygen (PIO₂). At around 19,700 feet (6,000 meters) there is less than half the atmospheric pressure than at sea level, and so about half of the number of oxygen molecules in the same volume of air. At around 33,000 feet (10,000 meters) the number of oxygen molecules in that same volume is halved again. This significant drop in atmospheric pressure makes it difficult for oxygen to cross the alveolar membrane and attach to passing red blood cells.

Altitude illness becomes increasingly common above 8,000 feet (2500 meters) and the incidence increases the more rapid the ascent. The most common altitude where you may encounter severe altitude illness is between 11,500 and 18,000 feet (5300-5500 meters). Altitudes above 18,000 feet are considered extreme and require special planning and equipment. In fact, there is so little oxygen available that the body begins to deteriorate rather than adapt.
Everyone reacts to being exposed to high altitude. If you were to take someone from sea level and drop them off at the summit of Mt. Everest at 29,000 feet (8800 meters) they would pass out within a few minutes and die shortly thereafter. However, climbers can ascend over a period of weeks without oxygen because the body can slowly adjust to the decrease in oxygen pressure. This increased tolerance of hypoxia is called acclimatization. The severity of hypoxic stress, the rate of ascent, and individual physiology will determine if your body will successfully acclimate.
Altitude-related syndromes are directly caused by the low pressure of the atmosphere and the resulting low concentration of oxygen. This is called hypobaric hypoxia or hypoxic hypoxia. There are many different types of high-altitude syndromes all with overlapping symptoms. For simplicity, altitude illness is grouped into three major syndromes:
- Acute Mountain Sickness (AMS),
- High Altitude Pulmonary Edema (HAPE), and
- High Altitude Cerebral Edema (HACE).
Acute Mountain Sickness (AMS)
AMS has been known about for hundreds of years, but thanks to rapid transportation more people are being exposed to high altitude without proper acclimatization. Even though AMS is the mildest of the three illnesses, the severity is increased with the rate of climb and the altitude gained. AMS typically starts at about 6,000 ft and gets worse as you go. There is little difference between men and women, level of physical fitness, or level of hydration in terms of susceptibility to AMS. However, obesity seems to increases your risk along with smoking and alcohol consumption.
Presentation
AMS will generally present in the setting of rapid ascent of unacclimatized people. The cardinal sign of AMS is a temporal or a non-focal headache that worsens with strenuous activity, especially bending over or with a Valsalva maneuver, overnight, or with increasing altitudes. Headaches are commonly followed by fatigue, dizziness, and loss of appetite. Most people describe these patient present as “sucking wind”.
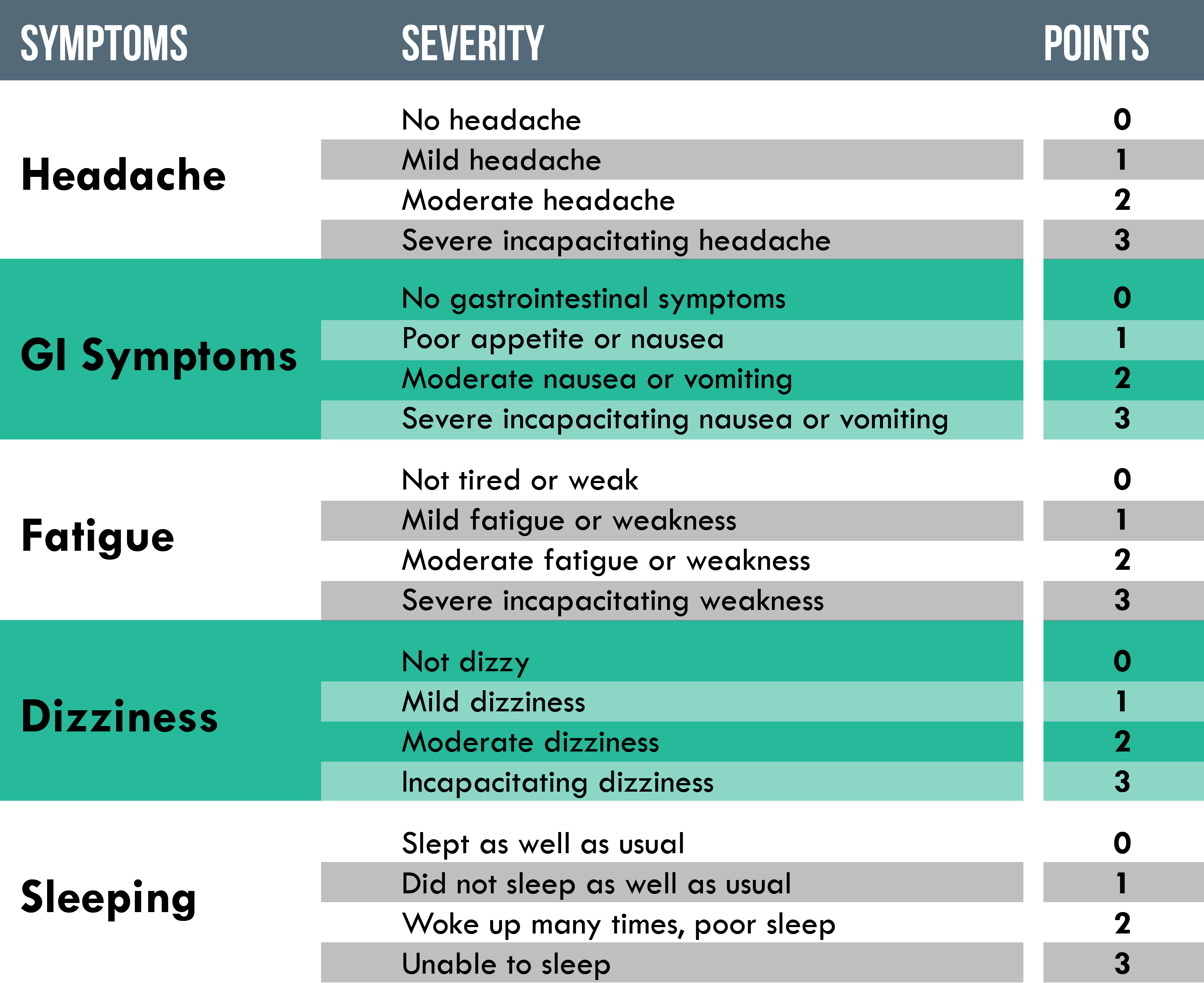
Since the symptoms of AMS are very similar to a hangover, dehydration, or viral infection it is often missed by inexperienced providers. Suspicion for AMS must go up in the setting of altitude without excluding other potential causes. Uncomplicated AMS doesn’t usually include a fever or aches and pains. To help facilitate this diagnosis the provider can utilize the Lake Louise Scoring System (LLSS). If the patient has a score of 3 or more, they are likely to have AMS.
Treatment
The best treatment for AMS is early recognition and allowance of acclimatization. Descent is the fastest way to resolve symptoms but may not be possible given the nature of the mission. If you hold position you must closely monitor the patient for progression of symptoms. If the symptoms remain or worsen despite 24 hours of rest and acclimatization you must descend. If any neurological or respiratory symptoms appear, you should immediately descend.
- Giving a nonsteroidal anti-inflammatory drug (NSAID) such as Aspirin or Ibuprofen (Motrin) and a pain reliever like Acetaminophen (Tylenol) may help combat the patients’ headache and help them descend.
- Consider treating the patient’s nausea with a non-drowsy antiemetic such as Ondansetron (Zofran).
- Low-flow supplemental oxygen is very effective down to 0.5-1 Lpm via nasal cannula.
- Portable hyperbaric chambers such as a Gamow bag are effective without supplemental oxygen but are generally not needed.
- The use of diuretics such as Acetazolamide (Diamox) or Furosemide (Lasix) to treat AMS has been described but not deeply studied and is usually not necessary.
AMS can best be prevented with proper acclimatization and a planned ascent profile. Some physicians may write prophylactic prescriptions for Diamox even acetazolamide has never formally been studied for altitude illness prevention. Even with appropriate preventive measures, individual physiological differences is the biggest factor determining whether someone will experience AMS. Being able to adjust the rate of ascent based on the most susceptible person will prevent AMS from progressing into something more dangerous.

High Altitude Pulmonary Edema (HAPE)
High altitude pulmonary edema (HAPE) is a non-cardiogenic edema that can affect people who ascend to altitudes 8,000 feet or higher and can happen even to acclimatized people. HAPE is the most common cause of death related to high altitude and is completely and easily reversed if recognized early and treated properly. The frequency of HAPE depends on many of the same factors as AMS, and may also include how cold it is or certain preexisting medical conditions, but is mostly determined by the rapid push to a sufficiently high altitude relative to where they started. The signs and symptoms of AMS are present in about half of HAPE cases.
Presentation
The early signs and symptoms of HAPE are decreased work performance and increased recovery time. The patient will display some of the same fatigue, weakness, and lassitude as with AMS, but may also have dyspnea on exertion and a dry or non-productive cough. The patient’s lips and fingertips may become cyanotic and they may become tachycardic and tachypneic. Increasing respiratory distress is the red flag for HAPE and the signal that the patient is has a potentially life-threatening condition.
HAPE usually occurs within the first 2 to 4 days of ascent to higher altitudes. However, HAPE may strike abruptly especially in a sedentary person who may not notice the early stages. Pink or blood-tinged, frothy sputum is a very late finding. Unlike in AMS, an elevated temperature as high as 101.3° F may be seen. Rales may be heard on one or both sides.
The pathophysiology of HAPE is a hypoxic pulmonary vasoconstriction (HPV) that causes leaky alveolar capillaries. HPV is a natural physiologic response to hypoxia. Blood flow is redirected from poorly ventilated lung regions to areas of better ventilation. The goal of this mechanism is to increase the delivery of oxygen to the blood. HPV is controlled by the detection of low oxygen levels in the endothelium of the lungs and is initiated by the smooth muscle cells. With prolonged hypoxia and HPV proper cellular respiration cannot take place. This constant constriction leads to pulmonary hypertension, and ultimately, pulmonary edema.
Also, recognize that this person is in respiratory acidosis. Respiratory acidosis is defined by alveolar hypoventilation in which there is an increase in partial carbon dioxide (PaCO) levels (hypercapnia.) This increase in PaCO2 is what causes the HAPE patient to be tachypneic. The respiratory compensatory system to increase the respiratory rate to off-gas as much CO2 as possible and recruit more O2. For the person at altitude, this is virtually impossible due to the mechanisms described above. This compensatory mechanism can only last so long with the given conditions. As the patient moves into a longer exposure, signs, and symptoms will increase. The patient will begin to have a productive cough containing pink frothy sputum, rales will become more widespread, you may hear audible gurgling, and their mental status will diminish.
Through the increased excretion of bicarbonate in the urine (bicarbonate diuresis), free-floating hydrogen ions in the blood will accumulate, causing a metabolic acidosis. The physiologic response to metabolic acidosis is for the patient to increase and deepen their respiratory rate to augment oxygenation.
Treatment
Treatment choices for HAPE depend on the severity of illness and mission considerations. As with all high-altitude illnesses, early recognition vastly improves the likelihood of successful outcome. In an austere setting, patients with HAPE need to be urgently evacuated to lower altitudes while minimizing their exertion.
- Keep the patient warm to prevent cold stress increasing pulmonary pressures.
- Attempt to decrease altitude by at least 1,600-3,200 feet (500-1000 meters). This can be done using a portable hyperbaric chamber like the Gamow bag while preparing for evacuation. Keep in mind that once you put someone in a Gamow bag, fresh air must be continuously cycled through using the foot pump. The person on the pump shouldn’t be the medic. Transportation while in a Gamow bag is nearly impossible because of the need for the foot pump.
- If evacuation isn’t immediately possible, the use of high-flow oxygen, 4 Lpm or higher, may temporize the symptoms. Use of a positive airway pressure technique (pursed-lip breathing) can be used for short periods, or a PEEP device can be used with or without a bag-valve-mask.
- Calcium channel blockers such as Nifedipine (Procardia) 30mg Extended Release every 12 hours (q12h) can be used to reduce pulmonary pressures. Although Nifedipine is used to lower blood pressure in some patients, it is unlikely to cause significant hypotension in healthy persons and works better than some other modalities (diuretics or vasodilators).
- Corticosteroids such as Dexamethasone (Decadron) can be used both as treatment and as prophylaxis. Dexamethasone has many actions in the lungs and the exact mechanism in play is not well understood. Dexamethasone will be discussed more in HACE below.
- Phosphodiesterase-5 Inhibitors such as Sildenafil (Viagra) 50mg every 8 hours (q8h) or Tadalafil (Cialis) 10mg every 12 hours (q12h) block hypoxic pulmonary hypertension as a side effect and therefore will prevent HAPE if taken at the first signs or symptoms. However, these medications may not be practical to order through common supply channels, sorry gents.
Prevention of HAPE begins with proper time for acclimatization, strict adherence to ascent timelines, and prophylactic use of Acetazolamide (Diamox) prior to the mission as discussed above.

High Altitude Cerebral Edema (HACE)
High-altitude cerebral edema (HACE) is an uncommon but deadly condition usually occurring in patients with AMS or HAPE. HACE is caused by two edematous mechanisms known as vasogenic and cytotoxic edema. Vasogenic edema is the leaking of fluids and proteins through the blood-brain barrier causing the brain tissue to swell. Cytotoxic edema is the movement of extracellular water into the cell, leading to the swelling of cells.
Presentation
HACE is extremely fatal if left untreated so early identification and treatment are key. As swelling increases, your patient will exhibit signs and symptoms of increased intracranial pressure. The hallmarks of HACE are ataxic gait, severe lassitude, and altered mental status including confusion, drowsiness, stupor, or coma. Headache, nausea, and vomiting occur frequently but are not always present. Hallucinations, cranial nerve palsy, paralysis of one side of the body, and seizures are also possible but uncommon. Progression from mild AMS to unconsciousness may be as rapid as 12 hours but usually requires a few days; less if the patient has HAPE as well.
Treatment
- Descent is the number one priority and treatment for this patient. Evacuation should be initiated at the first sign of ataxia or change in mental status. The use of a portable hyperbaric chamber is appropriate as discussed in HAPE.
- Corticosteroids such as Dexamethasone (Decadron) can be used to control brain swelling inside the skull. It is given as an initial dose of Dexamethasone (Decadron) 8mg PO, IM, or IV followed by 4mg every 6 hours (q6h)
- Supplemental high-flow oxygen at 4 Lpm or higher can be applied if available.
- Consider controlled hyperventilation while preparing other treatments (1 breath every 3 seconds or ventilate to an end-tidal CO2 of 30-40) via a bag valve mask may buy your patient time. Cerebral blood flow is highly dependent on PaCO2 levels. By hyperventilating your patient you can decrease PaCO2 and cause an cerebral arterial vasoconstriction temporarily. This technique is not a definitive treatment and can lead to a rebound increase in ICP caused by a sudden return to cerebral normocapnic levels from a hypocapnic state.
- Loop diuretics such as Furosemide (Lasix) 40-80mg IV or Bumetanide (Bumex) 1-2mg IV have been used to pull fluid from the brain, but you must be very cautious to maintain adequate fluid levels in the body.
- Hypertonic solutions such as hypertonic saline or Mannitol have been suggested but are not commonly carried by field providers.
How Do I Prevent Altitude Illness?
Acclimation is a very important step and top priority in preventing altitude illness. The approach is to have a slow and controlled or graded ascent. Recommendations by civilian organizations suggest limiting ascent to 1,000 feet per day (300 meters) above 3000 feet. The Army recommends a gradual ascent of no more than 1,000 to 2,000 feet per day to an area of sleep. Rather than push to 6,000 feet in a few hours, take that elevation over a few days. Not taking a calculated approach increases your risk of developing an illness.
Sleeping at altitude also means you are going to eat at altitude. As with any camping, there is nothing better than a hot meal. Caution must be taken when using a cooking stove in an enclosed space due to the risk of carbon monoxide (CO) poisoning from the emitted gases. CO has a much stronger affinity for hemoglobin than oxygen. It binds to hemoglobin to form carboxyhemoglobin, taking place of oxygen. CO poisoning closely mimics the signs and symptoms of acute mountain sickness to include, headache, confusion, and dizziness. This can make the diagnosis difficult, so remember to gather a strong history from this patient and begin high-flow oxygen as soon as possible.
Summary
- Although the mechanisms for each illness differ slightly, the main culprit is hypoxia. Stay ahead of the hypoxic power curve as best as you can with proper acclimation and prophylaxis.
- AMS begins as soon as your patient is experiencing a headache.
- Descent is the definitive treatment for each altitude illness.
- CO poisoning can be deadly and hard to detect, take all necessary precautions to limit CO exposure in small spaces.
Check on Learning
Use the following scenario to answer the questions below:
Your group is climbing a 14,001 foot peak. Your group consists of 29 people 24 men, 5 women, between 18 and 34 years of age, with good physical fitness. You know that about half the group uses tobacco, and about half regularly drinks alcohol. The mission is behind schedule and your leadership wants to make a push from base-camp at 3500 feet, past Camp I at 5200 feet, to Camp II at 7700 feet by the end of the day.
- What is the most likely medical outcome of the stated plan? What is the most dangerous medical outcome?
- How will your identified medical problems effect the mission?
- What are four ways you can reduce the likelihood of your identified medical problems? Which one is the best option for accomplishing the mission?
Scenario continued: You are at Camp II (7700 feet) and have six patients. Four with headaches and weakness and two with difficulty breathing. Some poor weather is limiting visibility to about ten feet. You are unable to call for evacuation by helicopter. You have a Gamow bag, and are carrying medications common to the mountain environment.
- What is wrong with your patients?
- What will your treatment plan be? How much medication will you need for six hours? 12 hours?
- What is your evacuation priority and why?
References
- Butler GJ, Al-Waili N, Passano DV, et al. Altitude mountain sickness among tourist populations: a review and pathophysiology supporting management with hyperbaric oxygen. Journal of medical engineering & technology. https://www.ncbi.nlm.nih.gov/pubmed/20836748. Accessed October 21, 2017.
- Hyperbaric Technologies Inc. Training and Reference Manual. January 2011.“The First Portable Hyperbaric Chamber for the Treatment of Acute Mountain Sickness”
- Fagenholz PJ, Gutman JA, Murray AF, Harris SN. Treatment of High Altitude Pulmonary Edema at 4240 m in Nepal. HIGH ALTITUDE MEDICINE & BIOLOGY . 2007;8. doi: 10.1089/ham.2007.3055
- Dehnert C, Mereles D, Greiner S, et al. Exaggerated Hypoxic Pulmonary Vasoconstriction Without Susceptibility to High Altitude Pulmonary Edema. High Altitude Medicine & Biology. 2015;16(1):11-17. doi:10.1089/ham.2014.1117.
- Luks AM, McIntosh SE, Grossom CK, et al. Wilderness Medical Society Practice Guidelines for the Prevention and Treatment of Acute Altitude Illness: 2014 update. Wilderness & Environmental Medicine. 2014;21(4):383-384. doi:10.1016/j.wem.2010.04.001.
- Tsurufuji S, Kurihara A, Ojima F. Mechanisms of anti-inflammatory action of dexamethasone: blockade by hydrocortisone mesylate and actinomycin D of the inhibitory effect of dexamethasone on leukocyte infiltration in inflammatory sites. The Journal of pharmacology and experimental therapeutics. https://www.ncbi.nlm.nih.gov/pubmed/6707940. Published April 1984. Accessed October 15, 2017.
- Hackett PH, Roach RC. High-Altitude Illness — NEJM. New England Journal of Medicine. http://www.nejm.org/doi/full/10.1056/NEJM200107123450206. Accessed October 12, 2017.
- Paralikar SJ, Paralikar JH. High-altitude medicine. Indian Journal of Occupational and Environmental Medicine. 2010;14(1):6-12. doi:10.4103/0019-5278.64608.
- Bartsch, P. Physiological aspects of high-altitude pulmonary edema. Journal of Applied Physiology, 98(3), 1101-1110. doi:10.1152/japplphysiol.01167.2004
- Baniya S, Holden C, Basnyat B. Reentry High Altitude Pulmonary Edema in the Himalayas. High Altitude Medicine & Biology. 00(00). doi: 10.1089/ham.2017.0088.
- Luks AM, McIntosh SE, Grossom CK, et al. Wilderness Medical Society Practice Guidelines for the Prevention and Treatment of Acute Altitude Illness: 2014 update. Wilderness & Environmental Medicine. 2014;21(4):383-384. doi:10.1016/j.wem.2010.04.001.
- Dunin-Bell O, Boyle S. Secondary Prevention of HAPE in a Mount Everest Summiteer. High Altitude Medicine & Biology. 2009;10(3):293-296. doi:10.1089/ham.2008.1094.
- FM 3-97.61 Military Mountaineering (2002)
- Cushing, T. A., Auerbach, P. S., & Harris, N. S. (2017). Auerbachs wilderness medicine. Philadelphia, PA: Elsevier.



Another great article by Ricky
LikeLike
Hi great readding your post
LikeLike