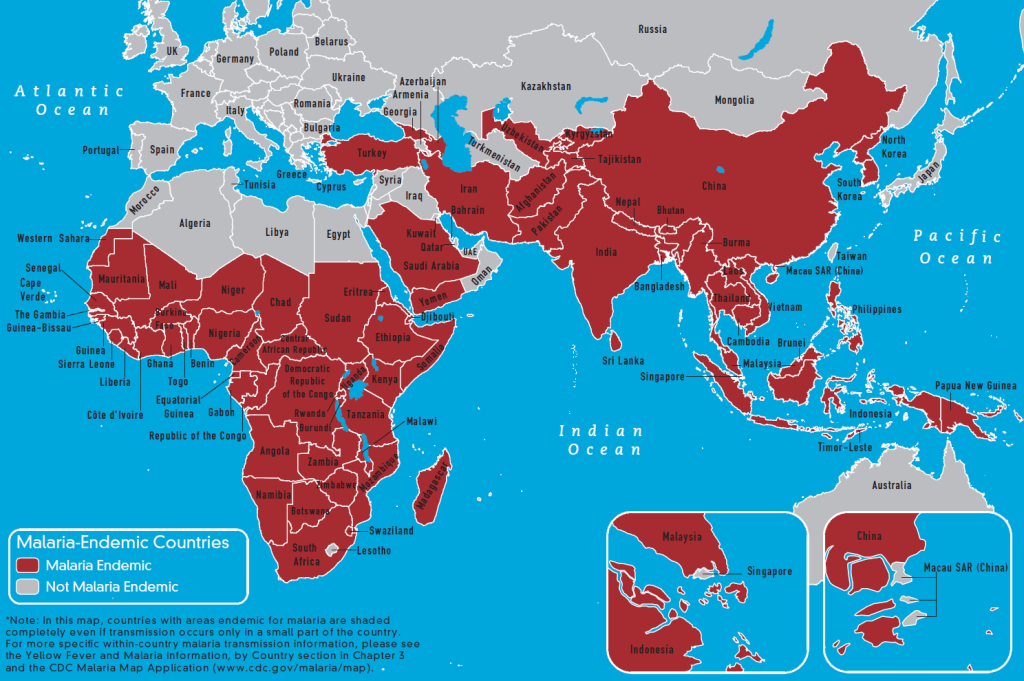
In 2018, malaria-infected an estimated 228 million people, killing over 400,000, disproportionately affecting the young and the poor (World Health Organization, 2019). Malaria is endemic to tropical and subtropical regions of the world, including parts of sub-Saharan Africa, Southeast Asia, and Central and South America (Suh, 2004). In 2003, the World Bank estimated that the disease is responsible for $12 billion U.S. dollars in economic revenue each year (Chima, 2003).
The World Health Organization estimates that 3.3 billion people, or approximately half the world’s population, are at risk of being infected with malaria (2019). Although methods of preventing transmission through individual and local protective measures are well-established, the disease is unlikely to be eliminated from endemic regions without some form of vaccine (Coelho, 2017). The development of an effective vaccine conferring long-term immunity to the malaria-causing parasite is a global scientific priority.
Life Cycle of Plasmodium
The disease-causing protozoan Plasmodium, is an obligate parasite in the family plasmodiidae, order haemospororida, class aconoidasida, and phylum apicomplexa. Apicomplexa is one of nine phyla in the “infrakingdom” or “superphylum” alveolate within the domain eukarya (Crutcher, 1996). One of the unique characteristics of apicomplexa parasites is their requirement of two hosts to complete their lifecycle. Five species of Plasmodium can infect humans: P. falciparum, P. knowlesi, P. vivax, P. ovale, and P. malariae. However, P. falciparum, P. ovale, P. vivax are the most commonly disease-producing (Talapko, 2019).
Plasmodium and the malarial disease it causes has been with us throughout recorded history. The first records of malarial diseases were found in Chinese medical writings dated back to 2700 BCE (Talapko, 2019). Some historians propose that Alexander the Great may have died of malaria in 323 BCE (Cunha, 2004; Sallares, 2004).
Clinically, the stages of Plasmodium life cycles can be divided into erythrocytic and pre-erythrocytic. However, it may be prudent to understand the complete lifecycle of the parasite to identify potential avenues of vaccination. The development of Plasmodium may be considered in four stages: (1) sexual reproduction within the vector or “mosquito stage”; (2) infection and development within the host’s liver or “pre-erythrocytic stage”; (3) infection and spread within the host’s red blood cells “erythrocytic stage”; and (4) development of gametocytes and transmission back to the vector or “sexual stage” (Crutcher, 1996).

Mosquito Stage
Plasmodium sporozoites spread through the saliva of the pregnant female Anopheles mosquito (Mahmoudi, 2018). The normally nectarivorous mosquito takes a blood meal to provide their offspring with proteins and amino acids not found in plant nectar.
Once in the mosquito’s midgut, the Plasmodium gametocytes undergo gametogenic meiosis to form Plasmodium eggs and sperm. Once fertilized, the resulting zygote undergoes mitosis to become a motile ookinete. The ookinete crosses the gut epithelium to form an oocyst outside the gut (Mueller, 2010) Following sporogony immature sporozoites are released into the mosquito’s body cavity and permeate the salivary glands to mix with saliva. The process of sexual reproduction and sporogony may take two to three weeks, and a female mosquito must live at least this long to transmit sporozoites to a human host (Crutcher, 1996).
Upon insertion of the feeding proboscis, the mosquito regurgitates infected saliva containing compounds that temporarily inhibit clotting, platelet aggregation, and inflammatory response while producing a local vasodilatory effect (Ribeiro, 2000). The mechanisms of these salivary compounds help defeat elements of human innate immunity and allow the immature sporozoites to enter the new host’s circulation quickly.
Liver (Pre-Erythrocytic) Stage
Following inoculation, the immature sporozoites enter into the systemic circulation (Vreden, 1994). Upon reaching the liver, the sporozoite tunnels through sentinel cells called Kupffer cells and into hepatic parenchyma to begin reproduction (Mazier, 1985). This process may occur in as little as 30 minutes from when the mosquito first bites (Crutcher, 1996).
Once inside a hepatocyte, the sporozoite undergoes a form of binary fission called schizogony. The result of schizogony is a schizont that contains many thousands of nuclei. The process of schizogony for malaria may take 5 to 21 days, depending on the species of Plasmodium, but may also enter a dormant state for several years. Once the mature schizont ruptures 2,000 to 40,000, mature merozoites are released back into circulation (Crutcher, 1996). Each merozoite is capable of infecting a single red blood cell.
Blood Stage
Hair-like surface proteins cover Plasmodium merozoites, allowing them to adhere to passing erythrocytes (Beeson, 2016). The attached merozoite invades the erythrocyte, shedding its protein coat and forming a schizont within the cell. The elapsed time between release into the bloodstream and invasion of an erythrocyte is between 30 and 90 seconds (Mohandas, 2012). Once inside the erythrocyte, the schizont produces more merozoites through schizogony over 48 to 72 hours.
In the species P. falciparum, merozoite invasion structurally alters the erythrocyte causing adherence to the endothelial lining of a blood vessel. If this adhesion occurs in the smaller vessels of the body, it may lead to occlusion of the vessel and subsequent anoxic tissue injury. Ischemia may be especially damaging in sensitive organs such as the brain, kidneys, or intestines leading to cerebral malaria, tubular necrosis, and intestinal ischemia and necrosis (Crutcher, 1996).
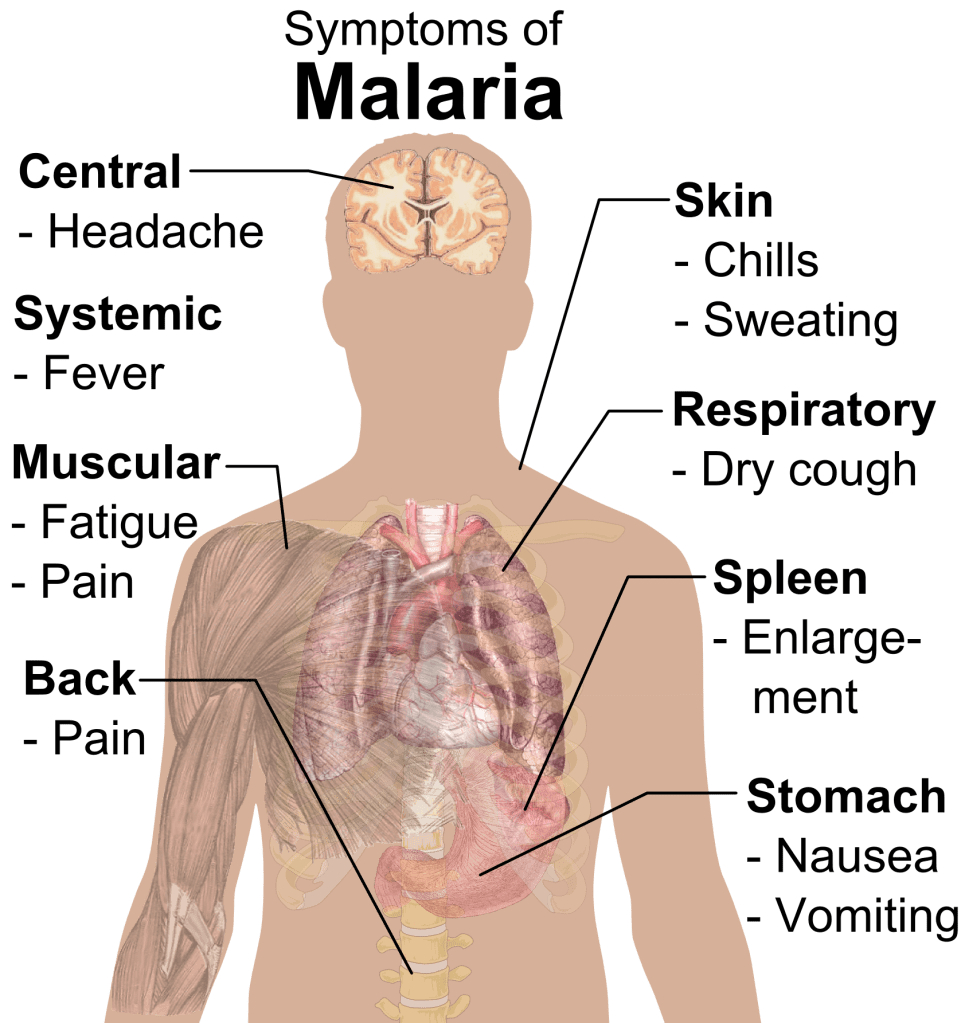
Asexual replication of merozoites culminates with the rupture of the red blood cell, releasing 20 to 40 merozoites. This pattern of erythrocyte invasion, replication, and rupture creates a pattern of symptoms with a periodicity between 48 and 72 hours. As red blood cells rupture in sufficient quantities, the victim feels chills and fever. As more red blood cells rupture, the human host becomes increasingly anemic. Those with severe or complicated malaria may experience any combination of respiratory distress, seizures, or shock (Suh, 2004).
Human hosts without previous exposure exhibit more severe symptoms. Children under the age of five years are especially at risk. Previous exposure produces limited immune protection in subsequent exposures; however, the severity of symptoms tends to decline with repeated infection. Patients may also exhibit splenomegaly resulting from increased filtration of ruptured erythrocytes and, less frequently, hepatomegaly with jaundice from destruction of liver tissue (Suh, 2004).
Sexual Stage
At some point during the cycle of erythrocytic infection, merozoites begin forming either schizonts or gametocytes. Female anopheles mosquitoes consume male and female gametocytes during a blood meal, and the process of transmission repeats.
Current Methods to Combat the Spread of Malaria
Erythrocytes may have naturally occurring genetic anomalies, which reduce the ability for a merozoite to attach and invade the cell. These genetic anomalies are sometimes in the form of modified surface proteins (Mohandas, 2012). However, sickle cell disease is, by far, the most common genetic defense against malaria (Crutcher, 1996). Of course, sickle cell anemia comes with its own set of complications and does not represent a reasonable approach to vaccination.
Methods of controlling malaria transmission involve many potential paths of transmission and generally include the destruction of breeding habitat, physical barriers between vector and host, chemical barriers such as insecticides (Tizifa, 2018). It is common for travelers to take chemoprophylactic medication before and while in malaria endemic regions. However, in recent years there have been increasing reports of chemoprophylaxis-resistant species (Talapko, 2019).
Malaria Vaccine Development
An array of biological, technological, economic, and social factors have delayed the development of a malaria vaccine (Mons, 1997). In addition to the complex, multi-host lifecycle already discussed, each stage and each species has different presenting antigens, sometimes with multiple forms. Hosts may be infected with multiple species simultaneously. The parasite effectively evades the human immune response and alters B-cell memory to limit the durability of acquired immunity (Mahmoudi, 2018).
Successes in Malaria Vaccine Development
Early scientific attempts at vaccination started in 1967. There have been many potential malaria vaccines brought to early clinical trials (Graves, 2006, 1; Graves, 2006, 2; Coelho, 2017; Draper, 2018). The effectiveness and duration of protection determine the success of a vaccine. Attaining 75% efficacy over two years is considered a crowning achievement (Draper, 2018).
Radiation-attenuated sporozoites (RAS) showed promise in rodents but failed to achieve adequate protection in humans (Draper, 2018). Vaccines that used viruses to target parasitized liver cells reached phase II clinical trials before being deemed non-efficacious. The initially promising SPf66 vaccine, which targeted the sporozoite stage, was only able to achieve a 30% efficacy during initial human trials (Graves, 2006, 1).
The most promising and, therefore, extensively tested candidate is the RTS,S/AS01 vaccine for P. falciparum (Graves, 2006, 2). RTS,S is classified as a sporozoite subunit vaccine and is designed to direct an immune response against the surface proteins covering the sporozoite while it travels from the site of inoculation. RTS,S achieved a 51% reduction in cases and conferred protection for around five years (Draper, 2018).
The Future of Malaria Vaccines
The Plasmodium organism has many innate characteristics which allow it to circumvent the human immune system. A key factor for vaccine success is the maintenance of protection after immunization (Coelho, 2017). The literature continues to support the pre-erythrocytic stage as the most promising window of opportunity for therapeutic intervention. The future of vaccine research will likely involve the improvement of current vaccines and the development of adjuvants, which boost immune response and memory.
References
- Beeson, J.G., Drew, D.R., Boyle, M.J., Feng, G., Fowkes, F.J.I., & Richards, J.S. (2016). Merozoite surface proteins in red blood cell invasion, immunity and vaccines against malaria. FEMS Microbiology Reviews, 40(3), 343–372. doi:10.1093/femsre/fuw001
- Chima, R.I., Goodman, C.A., & Mills, A. (2003) The economic impact of malaria in Africa: a critical review of the evidence. Health Policy, 63(1), 17-36. doi:10.1016/s0168-8510(02)00036-2
- Coelho, C.H., Doritchamou, J.Y.A., Zaidi, I., & Duffy, P.E. (2017). Advances in malaria vaccine development: Report from the 2017 malaria vaccine symposium. Nature Partner Journals Vaccines, 2(1) doi:10.1038/s41541-017-0035-3
- Crutcher, J.M., & Hoffman, S.L. (1996). Malaria. In Medical Microbiology (4th ed.). Galveston, TX: University of Texas Medical Branch. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK8584/
- Cunha, B.A. (2004) The death of Alexander the Great: malaria or typhoid fever? Infectious Disease Clinics of North America, 18(1), 53-63. doi:10.1016/S0891-5520(03)00090-4
- Draper, S.J., Sack B. K., King, C.R., et al. (2018). Malaria vaccines: Recent advances and new horizons. Cell Host & Microbe, 24(1), 43-56. doi:10.1016/j.chom.2018.06.008
- Graves, P. M., & Gelband, H. (2006). Vaccines for preventing malaria (SPf66). Cochrane Database of Systematic Reviews. doi:10.1002/14651858.cd005966
- Graves, P. M., & Gelband, H. (2006). Vaccines for preventing malaria (pre-erythrocytic). Cochrane Database of Systematic Reviews. doi:10.1002/14651858.cd006198
- Mahmoudi, S., & Keshavarz, H. (2018) Malaria vaccine development: The need for novel approaches: A review article. Iranian Journal of Parasitology, 13(1), 1-10.
- Mazier, D., Beaudoin, R., Mellouk, S., Druilhe, P., Texier, B., Trosper, J., et al. (1985). Complete development of hepatic stages of Plasmodium falciparum in vitro. Science, 227(4685), 440–442. doi:10.1126/science.3880923
- Mohandas, N., & An, X. (2012). Malaria and human red blood cells. Medical Microbiology and Immunology, 201(4), 593–598. doi:10.1007/s00430-012-0272-z
- Mons, B. (1997). The problem of multiplicity in malaria vaccine development. Annals of Tropical Medicine And Parasitology, 91(2), 55-58. doi:10.1080/00034989761300
- Mueller, A.-K., Kohlhepp, F., Hammerschmidt, C., & Michel, K. (2010). Invasion of mosquito salivary glands by malaria parasites: Prerequisites and defense strategies. International Journal for Parasitology, 40(11), 1229–1235. doi:10.1016/j.ijpara.2010.05.005
- Ribeiro, J. M. C. (2000). Blood-feeding in mosquitoes: probing time and salivary gland anti-haemostatic activities in representatives of three genera (Aedes, Anopheles, Culex). Medical and Veterinary Entomology, 14(2), 142–148. doi:10.1046/j.1365-2915.2000.00227.x
- Sallares, R., Bouwman, A., & Anderung, C. (2004). The Spread of Malaria to Southern Europe in Antiquity: New Approaches to Old Problems. Medical History, 48(03), 311–328. doi:10.1017/s0025727300007651
- Suh, K.N., Kain, K.C., & Keystone, J.S. (2004) Malaria. Canadian Medical Association Journal. 170(11), 1693-1702. doi:10.1503/cmaj.1030418
- Talapko, J., Škrlec, I., Alebić, T., Jukić, M., & Včev, A. (2019) Malaria: The past and the present. Microorganisms. 7(6), 1-17. doi: 10.3390/microorganisms7060179
- Tizifa, T.A., Kabaghe, A.N., McCann, R.S., van den Berg, H., Van Vugt, M., & Phiri, K.S. (2018). Prevention Efforts for Malaria. Current Tropical Medicine Reports, 5(1), 41–50. doi:10.1007/s40475-018-0133-y
- Vreden, S. G. S. (1994). The role of Kupffer cells in the clearance of malaria sporozoites from the circulation. Parasitology Today, 10(8), 304–308. doi:10.1016/0169-4758(94)90084-1 World Health Organization. (2019). World malaria report 2019. Retrieved from https://www.who.int/publications-detail/world-malaria-report-2019

